Cytogenetic Chromosomal Abnormalities
Authors
INTRODUCTION
Chromosomal duplication, pairing, and disjunction are important events preceding the transmission of genetic material during cellular mitoses. Errors resulting during these steps have dramatic consequences, especially when they occur at meiosis, fertilization, and early cleavage divisions. Most numeric chromosomal abnormalities are incompatible with life and have an incidence of 50% in spontaneous abortions, with trisomies for the chromosomes 21, 13, 18, and 16 and gonosomes representing approximately 50% of chromosomally abnormal abortions.1,2,3,4
The risk of transmitting genetic disorders to the offspring has always been of great concern for couples contemplating pregnancy. In patients with a normal karyotype, numeric chromosome abnormalities are generated ex novo, and maternal age is the only recognized risk factor for aneuploidy. In the last decades, the evaluation of the family history and age of the female partner in combination with prenatal diagnosis has substantially decreased the incidence of affected pregnancies ongoing beyond 3 months’ gestation. The possibility of anticipating the diagnosis of the chromosomal condition to the preimplantation stage was proposed in the early 1960s and reached the first application in sexing rabbit blastocysts.5 In the following years, a great effort has been dedicated to define techniques and strategies in molecular biology to be applied to single cell analysis.6,7,8 This has made preimplantation genetic diagnosis (PGD) an established technique in specialized in vitro fertilization (IVF) laboratories, where it is offered as an additional tool to increase the rate of healthy pregnancies.
PREIMPLANTATION GENETIC DIAGNOSIS FOR ANEUPLOIDY
In 1993, the first results of the clinical application of PGD for aneuploidy in combination with IVF treatment cycles were reported.9 Following ovarian stimulation and IVF, embryos underwent blastomere biopsy and chromosomal analysis, with the aim of selecting the embryos for transfer on the basis of their chromosomal condition. The original idea was based on the observation that women in advanced reproductive age experience a decline in implantation. Although several factors could be implicated, the main cause is represented by an increased risk of mitotic errors at oogenesis whose immediate clinical consequences are spontaneous abortion and implantation failure. Therefore, it was postulated that the identification of euploid embryos could reverse the age effect, and PGD for aneuploidy has been applied not only to reduce the incidence of trisomic pregnancies but also to improve the ongoing implantation rate by decreasing the occurrence of spontaneous abortion. The results obtained have confirmed that the tendency to develop aneuploid embryos increases proportionally to maternal age.10,11,12 Data have also shown that the incidence of chromosomal abnormalities in preimplantation embryos was notably higher than that reported in spontaneous abortions, indicating a strong selection against aneuploidy at implantation. This is confirmed by the finding that monosomies, with the exception of monosomy X and 21, are undetected in clinical pregnancy, although they occur at the same frequency as trisomies. It is conceivable that, in agreement with animal studies and in vitro culture of human embryos, monosomies arrest developing following human genome activation.13,14 Accordingly, the rate of blastocyst formation has been reported to be dependent on maternal age, with a decrease at older ages.15
All these data provided the molecular support to the already known decline in the reproductive performance depending on age. Impulse was given to the application of PGD for aneuploidy in couples with a poor prognosis of pregnancy, and categories of patients were identified who yield high proportions of chromosomally abnormal embryos.12,16,17 In some of them, the selection of embryos for transfer based on PGD demonstrated an increased implantation rate and a concomitant reduction in the incidence of spontaneous abortions owing to the highest viability associated with a normal chromosomal complement.17,18
The Technique
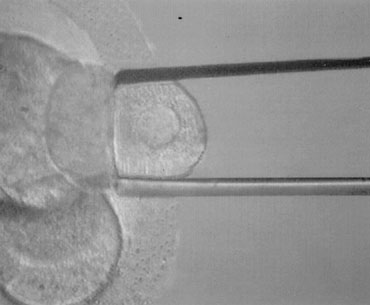
PGD is performed on blastomeres biopsied from regularly developing day 3 embryos by removing one or two cells. The effects of biopsy on embryo viability had been studied at different stages in the mouse. Embryo damage as the result of the procedure itself was negligible, and the highest live birth rates derived from biopsy done at the eight-cell stage compared with earlier stages, possibly due to the reduction in the embryonic mass (1/8 instead of 1/4). Embryo biopsy is generally carried out at 62 to 64 hours after insemination. At this time, compaction starts to take place, and cellular damage is very likely to occur if the biopsy is performed when cell-cell interactions and junctions begin to assemble. Alternatively, embryos can be shortly incubated in a divalent cation–deficient medium. The biopsy procedure entails an opening in the zona pellucida of approximately 20 to 25 μm diameter, which is performed chemically, mechanically, or using contact laser. The position of the embryo is selected to have a nucleated blastomere at the three o’clock position that is removed by using a polished glass needle and gently released in the medium (Fig. 1). Extreme attention is taken to avoid rupture of the cell membrane and damage to the surrounding blastomeres. The biopsied embryo is removed from the biopsy disk, washed, and incubated in fresh medium, whereas the blastomere undergos genetic analysis.
|
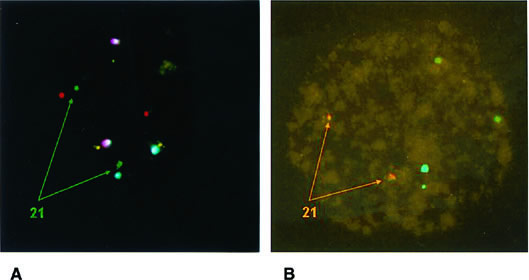
Conventional karyotyping is not applicable to PGD owing to the low metaphase yield and the long time necessary for all the steps involved. The technique of fluorescence in situ hybridization (FISH) permits chromosome enumeration to be performed in interphase nuclei and, with the use of multiple probes, provides the diagnosis of different chromosomes that can be completed by subsequent rounds with additional probes (Fig. 2). In this way, diagnosis of nine or more chromosomes is currently available, including those that are more frequently associated with abnormalities in clinical pregnancies: XY, 13, 15, 16, 18, 21, and 22.19,20,21 This is done in a time frame compatible with IVF, and embryo transfer is normally performed on day 4 or day 5 with the purpose of (1) having enough time to complete the genetic analysis; (2) selecting embryos on the basis of morphology and PGD results; and (3) reducing the risk of losing blastomeres through the breach opened in the zona pellucida during the transfer procedure owing to more advanced compaction.22
The high risk of developing aneuploid embryos as well as the error rate inherent in one-cell analysis should prompt the physician to advise pregnant patients to undergo conventional prenatal diagnosis to confirm and complete the results obtained by PGD.23
Fluorescence In Situ Hybridization: Scoring Criteria
The analysis performed on a single cell makes scoring criteria extremely important to enable a correct diagnosis. Basically, the distance between two signals specific for the same chromosomes must be at least two domains (a domain is represented by the diameter of a signal); if the distance is shorter, the pattern is considered to belong to split signals.24 Valuable information on FISH accuracy and for the formulation of scoring criteria derived from the analysis of all the blastomeres in nontransferred embryos.23,25 As represented in Table 1, 711 embryos were spread and their nuclei were fixed and analyzed with the same probes used for PGD. PGD results were confirmed in 91%; misdiagnosis occurred in 62 embryos but with a clinical relevance in only 31 (4%): 27 were erroneously classified as abnormal and 4 as normal; the remaining 31 carried an abnormality different from that diagnosed by PGD.
TABLE 1. Embryo Spreading and Fluorescence In Situ Hybridization Reanalysis of All Blastomeres*
Reanalysis† | ||||
PGD | Embryos (n) | Confirmed | Normal | Other Abnormality |
Normal | 137 | 133 (97) | — | 4 (3) |
Monosomic | 193 | 164 | 9 | 20 |
| One chromosome | 135 | 118 | 5 | 12 |
| Two chromosomes | 45 | 40 | 3 | 2 |
| Three chromosomes | 13 | 6 | 1 | 6 |
Trisomic | 184 | 164 | 14 | 6 |
| One chromosome | 166 | 151 | 12 | 3 |
| Two chromosomes | 15 | 11 | 1 | 3 |
| Three chromosomes | 3 | 2 | 1 | 0 |
Haploid | 19 | 17 | — | 2 |
Polyploid | 34 | 29 | 2 | 3 |
Complex abnormalities | 16 | 16 | — | — |
Total | 711 | 449 (91) | 27 (4) | 35 (5) |
PGD, preimplantation genetic diagnosis.
* Results are related to those obtained by PGD on one cell.
† Values in parentheses are percentages.
Different sources of FISH error have been identified: mosaicism, signal overlaps or splitting, failed hybridization, and loss of micronuclei during fixation. Mosaicism affects approximately 30% of in vitro–generated embryos, and the consequences for further development are diverse, depending on the type of mosaic and the proportion of abnormal cells.26 This condition is not efficiently diagnosed at PGD; however, the use of multiple probes enables the detection of complex abnormalities.
According to the data presented in Table 1, the highest source of error was represented by false monosomies. Consequently, adequate fixation protocols for the formation of good spreads have been proposed to minimize errors during this step owing to loss of micronuclei and signal overlapping.24,27 Improvement can also be derived by the use of a computerized image analysis system. A special strategy aimed at verifying failed hybridization and signal overlapping is based on the use of two probes for the same chromosome that target different loci. This is routinely done for chromosome 21, whose clinical relevance makes the use of two different probes a safety measure to prevent misdiagnosis (Fig. 3).28
Preimplantation Genetic Diagnosis on Polar Body
PGD relies on the analysis of both polar bodies to deduce the oocyte chromosomal status.29 The main advantage of the technique is the maintenance of embryo integrity owing to the fact that just meiosis by-products are used to infer the oocyte condition. However, the impossibility of diagnosing paternally derived defects and those originated after fertilization or the first cleavage events imposes notable limits to the application of this approach compared with embryo biopsy.
CLINICAL INDICATIONS
Between September 1996 and December 2001, PGD for aneuploidy was performed in 634 conception cycles at S.I.S.ME.R. Reproductive Medicine Unit. A diagnosis was obtained in 3262 embryos (99% of 3290 biopsied embryos); of these, 1042 had a normal chromosomal complement (32%) and made the transfer possible for 418 cycles (66%) in which an average of 1.8 ± 0.7 euploid embryos were replaced, yielding 128 clinical pregnancies (31%) and an implantation rate of 22.3%. The analysis of the data revealed that the proportion of aneuploid embryos is very high in some categories of patients with a poor prognosis for pregnancy. This could represent the main cause of reproductive failure.
Advanced maternal age has been the first indication for PGD of aneuploidy based on the hypothesis that the selection of chromosomally normal embryos could reverse the age effect. Other categories of younger patients were also included in the study: multiple unexplained failures of previous IVF cycles; an altered karyotype owing to the presence of aneuploid cell lines of sex chromosomes; a severe male factor in couples with at least one IVF failure; and the occurrence of repeated spontaneous abortions.
Advanced Maternal Age
The increase in aneuploidy rate according to maternal age resulting from pregnancies has been confirmed in embryos. As reported in Table 2, 327 cycles with a maternal age of 36 years or older and a normal karyotype underwent PGD. A total of 1717 embryos were FISH diagnosed, resulting in 498 chromosomally normal (29%), whereas the remaining 1204 (71%) carried chromosomal abnormalities that were not compatible with either implantation or a healthy pregnancy. Euploid embryos were transferred in 202 cycles, generating 62 clinical pregnancies (31%) and an implantation rate of 21.7%. Spontaneous abortion occurred in 7 cases (11%).
TABLE 2. Preimplantation Genetic Diagnosis for Aneuploidy: Results Divided According to Indications
≥36 Yr | ≥3 IVF Cycles | Altered Karyotype | |
Cycles (n) | 327 | 81 | 54 |
Age (y) (M ± SD) | 39.8 ± 2.6 | 32.3 ± 2.4 | 34.6 ± 3.6 |
Previous IVF cycles (n) (M ± SD) | 2.6 ± 2.1 | 4.0 ± 1.4 | 1.0 ± 1.3 |
Embryos FISH analyzed (n) | 1717 | 435 | 298 |
Embryos FISH diagnosed (n) | 1702 | 429 | 295 |
FISH normal (n[%]) | 498 (29) | 174 (41) | 109 (37) |
FISH abnormal (n[%]) | 1204 (71) | 255 (59) | 186 (63) |
Transferred cycles (n [%]) | 202 (62) | 62 (76) | 39 (72) |
Transferred embryos (n) (M ± SD) | 1.8 ± 0.8 | 2.1 ± 0.8 | 2.2 ± 0.8 |
Clinical pregnancies (n [%]) | 62 (31) | 20 (32) | 19 (49) |
Abortions (n) | 9* | 1† | 1 |
Implantation rate (%) | 21.7 | 21.7 | 33.7 |
FISH, fluorescence in situ hybridization; IVF, in vitro fertilization; M, mean; SD, standard deviation.
* Two ectopic pregnancies.
† One ectopic pregnancy.
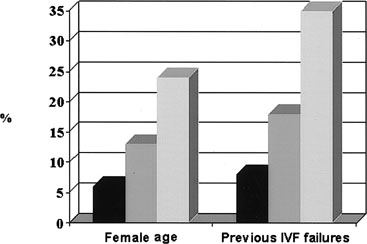
The proportion of chromosomal abnormalities increased proportionally to age, ranging from 64% in women ages 36 to 37 years to 86% detected in those aged 43 years and older. Pregnancy and implantation rates varied accordingly, with a significant decrease starting at 43 years (Fig. 4), implying that FISH selection reduces the effects of the age factor, providing the same reproductive performance to patients aged between 36 and 42 years.17 Other factors arise at older ages that may contribute to a reproductive decline; among them, a compromised uterine receptivity or the involvement in aneuploid events of chromosomes different from those tested.
|
Repeated In Vitro Fertilization Failures
Young patients with unexplained repeated IVF failures represent a challenge to IVF clinics. The idea of proposing PGD to them originated in the effort of identifying a possible cause of implantation failure.12,16 Detection of chromosomal abnormalities in 59% of the FISH-analyzed embryos indicated that the generation of numeric chromosomal defects could be one of the most important factors determining this poor prognosis condition. Table 2 reports the data yielded by 81 cycles; embryo transfer was performed in 62 cycles (76%), obtaining 20 clinical pregnancies (32%) and an implantation rate of 21.7%.
The analysis of the detected abnormalities contributed to characterize this category of patients that substantially differ from the group of maternal age patients. The most frequent defects were complex abnormalities, haploidy, and polyploidy, which globally accounted for 67% of total abnormalities compared with 55% in patients with advanced maternal age (p <.001; Table 3). The incidence of polyploidy was especially high (12%) and occurred at the same frequency after intracytoplasmic sperm injection (ICSI) or conventional IVF insemination.30 These findings denote alterations in the events or structures entering cell divisions such as asynchrony between karyokinesis and cytokinesis or centriolar defects. The male gamete could play an important role in determining this condition, taking into consideration that the sperm-derived centriole and corresponding microtubule-organizing regions precede the first oocyte divisions.31 In addition, the sperm centrosome controls the plane of the first cleavage with critical consequences for the polarity and ploidy of the resulting embryo.32 Interestingly, the proportion of couples with a male factor infertility was 67% in the group of repeated IVF failures (47% in the group of patients with advanced maternal age); this could support the results obtained from the FISH analysis. This observation suggests that this group is probably heterogeneous as far as the main cause of reproductive failure is concerned. More data need to be collected and properly analyzed to verify whether a series of concomitant factors or chromosomes different from those tested could contribute to this poor prognosis condition.
TABLE 3. Analysis of Chromosomal Abnormalities According to Preimplantation Genetic Diagnosis Indications
≥36 Yr | ≥3 IVF Cycles | Altered Karyotype | |
Embryos FISH diagnosed (n) | 1702 | 429 | 295 |
FISH abnormal (n [%]) | 1204 (71)*† | 255 (59)* | 186 (63)† |
Monosomy + trisomy (n [%]) | 537 (45)* | 85 (33)* | 79 (42) |
Haploid (n [%]) | 48 (4) | 13 (5) | 4 (2) |
Polyploid (n [%]) | 71 (6)† | 29 (12)† | 12 (6) |
Complex abnormalities (n [%]) | 548 (45) | 128 (50) | 91 (49) |
FISH, fluorescence in situ hybridization; IVF, in vitro fertilization.
*p < .001.
†p < .005.
†p < .025.
Altered Karyotype
The reproductive risk for carriers of gonosomal mosaicism (45,X0; 47,XXX; 47,XXY; 47,XYY) strictly depends on the presence of mosaic cell lines in the gonads. Although the clinical relevance of this condition is not clear, carriers of these aneuploid states are advised to undergo prenatal diagnosis in case of pregnancy. PGD has been proposed to these patients with the aim of (1) verifying the incidence of chromosomally abnormal embryos and (2) decreasing their reproductive risk by selecting euploid embryos for transfer.
Table 2 shows the results obtained by PGD applied to carriers of gonosomal mosaicism (54 cycles). After FISH analysis, 63% of the embryos showed an abnormal chromosomal complement. Monosomy and trisomy contributed 42% of total abnormalities, an incidence similar to that detected in the group of advanced maternal age (see Table 3). The rate of aneuploidy for sex chromosomes was 5.3% and not different from that characterized in the other PGD categories (2.6% in the group of maternal age and 4.7% in couples with IVF failures). The transfer of FISH-normal embryos was achieved in 39 cycles and yielded 19 clinical pregnancies (49%) with an implantation rate of 33.7%. Only 1 pregnancy miscarried, leading to a take-home-baby rate of 33%. These results suggest that these patients could have a predisposition to generate high rates of chromosomally abnormal embryos, probably due to disturbances in the mechanisms entering cell division that make them prone to yield different cell lines.
Microsurgical Sperm Aspiration—Testicular Sperm Extraction
The introduction of ICSI has made the treatment of patients with extremely severe male factor infertility very common in reproductive medicine, with a proportion of approximately 1:1 compared with conventional insemination. The follow-up of the children born after ICSI has demonstrated an increased incidence of de novo chromosomal abnormalities, with a notable rise in the aneuploidies accounting for sex chromosomes.33,34
In agreement with this finding, although no variation in the overall percentage of chromosomally abnormal embryos according to sperm parameters was detected, a higher incidence of monosomies and trisomies was found in embryos generated by sperm retrieved with microsurgical sperm aspiration (MESA) and testicular sperm extraction (TESE); in addition, the rate of gonosomal aneuploidy increased proportionally with the severity of the male factor condition.35
PGD for aneuploidy was proposed to azoospermic patients who had performed at least one conception cycle without getting an on-term pregnancy. The results, reported in Table 4, demonstrated that in both MESA and TESE patients the rate of chromosomally abnormal embryos was high (75% and 71%, respectively). Consequently, the proportion of transferred cycles was reduced (50% of the oocyte retrievals for MESA and 55% for TESE) yielding three clinical pregnancies in each category. The analysis of the detected abnormalities revealed that the chromosomal constitution of the generated embryos is significantly different between the two groups with a higher incidence of monosomy and trisomy in embryos generated by MESA (52% versus 34% in TESE; p <.05). Conversely, the proportion of complex abnormalities was superior in the TESE group (40% versus 22%; p <.05).
TABLE 4. MESA and TESE Patients With at Least One Previous In Vitro Fertilization Attempt
MESA | TESE | Total | |
Cycles (n) | 16 | 31 | 47 |
Age (y) (M ± SD) | 32.1 ± 4.3 | 35.8 ± 4.4 | 34.5 ± 4.7 |
Embryos FISH diagnosed (n) | 72 | 139 | 211 |
FISH normal (n [%]) | 18 (25) | 41 (29) | 59 (28) |
FISH abnormal (n [%]) | 54 (75) | 98 (71) | 152 (72) |
Monosomy-trisomy (n [%])* | 28 (52)† | 33 (34)† | 61 (40) |
Gonosome aneuploidy (n [%])* | 4 (7.4) | 6 (6.1) | 10 (6.6) |
Transferred cycles (n [%]) | 8 (50) | 17 (55) | 25 (53) |
Transferred embryos (n) (M ± SD) | 1.6 ± 0.7 | 1.8 ± 0.7 | 1.8 ± 0.7 |
Clinical pregnancies (n [%]) | 3 (38) | 3 (18) | 6 (24) |
Abortions (n) | 0 | 0 | 0 |
Implantation rate (%) | 30.8 | 12.9 | 18.2 |
FISH, fluorescence in situ hybridization; M, mean; SD, standard deviation; MESA, microsurgical sperm aspiration; TESE, testicular sperm extraction.
* Calculated over total abnormalities.
†p < .05.
Despite the high incidence of abnormalities, the clinical outcome after PGD was not excellent, especially in the case of TESE. The causes of these poor prognosis conditions are probably multifactorial and, as discussed previously, a significant involvement of the male gamete in the etiology of these alterations is probable. At confirmation of the data derived from the follow-up of the babies born after ICSI, the rate of gonosomal aneuploidy was quite high (6.6% versus 2.6% that was detected in the category of couples with an age factor; p <.05), with no significant differences between MESA and TESE (7.4% versus 6.1%).
In view of these results, which add some concern about the outcome of the ICSI offspring, the chromosomal analysis of the embryos generated by MESA and TESE could be considered as a possible approach in the framework of prevention in reproductive medicine.
Other Indications
Other categories with a poor prognosis of full-term pregnancy have been proposed to undergo PGD for aneuploidy with the aim of understanding whether the low implantation rate in these patients depends on a chromosomal imbalance. This approach has also been followed in the clinical application of new programs, such as oocyte freezing and in vitro maturation.
RECURRENT ABORTIONS.
Although this patient category is probably heterogeneous in its etiology, preliminary data derived by PGD report that these couples yield a high percentage of chromosomally abnormal embryos.36
The clinical application on a series of 80 cycles is shown in Table 5. According to FISH results, only 29% of the 435 analyzed embryos were chromosomally normal. Transfer was achieved in 54 cases with 20 clinical pregnancies (37% per transferred cycle and 25% per oocyte retrieval); 3 ended in spontaneous abortion, yielding a take-home-baby rate of 21.2% per oocyte retrieval. These data suggest that aneuploidy can represent the cause of repeated miscarriages, although other factors probably contribute to this condition. Additional data are required to verify this hypothesis, especially from young patients with no other poor prognosis indications such as maternal age, altered karyotype, or severe male infertility.
TABLE 5. Preimplantation Genetic Diagnosis for Aneuploidy in Patients With Repeated Abortions: Fluorescence In Situ Hybridization and Clinical Results
Cycles (n) | 80 |
Age (y) (M ± SD) | 37.6 ± 4.4 |
Embryos FISH analyzed (n) | 437 |
Embryos FISH diagnosed (n [%]) | 435 (99) |
FISH normal (n [%]) | 125 (29) |
FISH abnormal (n [%]) | 310 (71) |
Transferred cycles (n [%]) | 54 (67) |
Transferred embryos (n) (M ± SD) | (1.8 ± 0.7) |
Clinical pregnancies (n [%]) | 20 (37) |
Abortions (n) | 3 |
Implantation rate (%) | 26.0 |
FISH, fluorescence in situ hybridization; M, mean; SD, standard deviation.
POOR RESPONDERS.
Women with a poor response to hormonal stimulation demonstrated high percentages of chromosomally abnormal embryos (approximately 70%), implying that the great majority of the few embryos available are not compatible with healthy implantation.37 To evaluate the significance of this factor independently of female age, 137 PGD cycles with a maternal age of 35 years or younger were studied. Figure 5 represents the results obtained according to the number of retrieved oocytes that were arbitrarily divided in three categories: 1 to 3, 4 to 7, and 8 or higher. Probably owing to the small number of patients included in the first group, significant differences were not reached. However, there was a trend suggesting that in young, poor-responder women, the proportion of chromosomally normal embryos is extremely low (21%), with monosomy and trisomy contributing 45% of total abnormalities. If these data are confirmed, a strict correlation between ovarian environment and oogenesis could be postulated.
|
OOCYTE FREEZING.
Despite numerous efforts, embryos developed by thawed oocytes have a very poor implantation potential. PGD was performed on embryos generated by oocytes that were thawed after vitrification,38 with the aim of verifying whether the freezing procedure could affect the following divisions and generate aneuploidy. Seventeen embryos from 10 patients were studied; 9 were euploid and the remaining 8 carried chromosomal abnormalities. A regular embryo development was mostly associated with a normal chromosomal status: of 11 embryos with at least five cells on the morning of day 3, 7 were chromosomally normal, whereas slow cleaving or arrested embryos, a common condition in this program, exhibited complex abnormalities. Damage to the cytoskeleton and consequent alteration of the mitotic process could derive from the procedure of oocyte freezing. This could cause irregular division or, in the most severe cases, blockage in development.
OOCYTE In Vitro MATURATION.
The purpose of performing a chromosomal screening was to verify the incidence of aneuploidy with special regard to the frequent occurrence of slow cleavage and blockage in the growth of in vitro maturation (IVM) embryos. The analysis of arrested embryos demonstrated complex abnormalities with most of the chromosomes present in multiple copies.30 It can be argued that cytokinesis failed to occur after chromosome duplication, in agreement with the observation that IVM cannot support the acquisition of cytoplasmic maturation with direct consequences on the mechanisms involved in cell growth.39
PREIMPLANTATION GENETIC DIAGNOSIS FOR ANEUPLOIDY: A PROGNOSTIC VALUE
A retrospective analysis was performed on the data derived from patients who repeated PGD cycles at least once. The study included 114 couples who underwent 144 cycles after the first unsuccessful PGD attempt (Table 6).
TABLE 6. Repeated Preimplantation Genetic Diagnosis Cycles
| Euploid Embryos in the First PGD Cycle (n) | ||||
| 0 | 1 | ≥2 | Total | |
| First cycle | 29 | 27 | 58 | 114 |
| Second cycle | 6 | 5 | 14 | 25 |
| Fourth cycle | 0 | 3 | 2 | 5 |
| Total | 35 | 35 | 74 | 144 |
PGD, preimplantation genetic diagnosis.
In couples with an age factor, the same pattern of chromosomal abnormalities was maintained in the subsequent cycles, with monosomies and trisomies having the same incidence throughout, reaffirming that aneuploidy is strictly dependent on female age. Patients with no euploid embryos at the first PGD cycle had very low probability of having an on-term pregnancy in the following attempts (6% live-birth rate), whereas a 24% live-birth rate in subsequent attempts was associated with the detection of at least two euploid embryos in the first PGD cycle (Fig. 6). A similar trend was observed in couples whose indication for PGD was a history of 3 or fewer previous IVF failures. Despite a wider variability in the percentage of chromosomally abnormal embryos compared with those in patients with advanced maternal age, the best prognosis (35% live-birth rate) remained for those patients who had at least two chromosomally normal embryos at the first attempt versus an 8% live-birth rate related to the absence of chromosomally normal embryos in the first cycle (see Fig. 6).
The analysis of these data provides a sort of prognostic value on the basis of the general outcome that each couple experienced in its first PGD attempt. This is a valuable information regarding future expectations, which helps the experts of reproductive medicine in the difficult task of advising patients about the worthiness of repeating attempts with a reasonable hope of having children.
CHROMOSOME-SPECIFIC ANEUPLOIDY
Besides the clinical relevance of PGD for aneuploidy, this technique has also contributed novel information on early embryology. Two are especially relevant, namely (1) the association between ploidy and embryo morphology and (2) the frequency at which specific chromosomes give rise to aneuploid events.
A normal chromosomal complement is mostly associated with a regular morphology. Embryos with seven or eight cells at 62 hours postinsemination with less than 10% fragmentation, absence of multinucleation, or cytoplasmic concentration have the best chances (around 50%) of being chromosomally normal.40 Nevertheless, the tendency in patients with PGD indications to generate high rates of abnormalities does not preclude the possibility of aneuploidy, even in embryos developed to morphologically normal blastocysts.14,26,40 These findings suggest that morphologic criteria alone are of limited value in these patient categories.
One-cell diagnosis imposes strict limits on the number of chromosomes to be tested. Data from spontaneous abortions and live births were obviously taken into consideration, and the probes for the aneuploid chromosomes most commonly found were included in the hybridization mixture. However, the analysis of PGD data has started to bring some queries about the selection of chromosomes that are currently screened. It is now clear that the frequency at which chromosomes originate aneuploidies occurs at different rates for each chromosome. In the group with an age factor, in which segregation errors are more likely to occur, chromosomes 1, 15, 16, 17, 21, and 22 exhibited the highest rate of variation (Table 7); there was a proportional increase in relation to age for the chromosomes 15, 21, and 22 (Fig. 7), chromosomes 1 and 17 were significantly lower in the youngest categories, whereas chromosomes 13, 14, 16, 18, X, and Y showed similar aneuploidy rates in all age groups.25,30 These data, extended to other chromosomes as well as to younger women, could provide valuable information on the chromosomes that are more prone to variation in the different age groups. It would also be possible to verify whether chromosomes exist whose numeric variations have such a deleterious effect on embryo viability to make this condition completely incompatible with implantation.
TABLE 7. Distribution of Chromosomal Numeric Abnormalities per Single Chromosome in Relation to Preimplantation Genetic Diagnosis Indications
| Chromosomes | ≥36 Yr (%) | ≥3 IVF Cycles (%) | Altered Karyotype (%) | MESA-TESE (%) |
| XY | 30 | 30 | 31 | 36 |
| 1 | 38 | 34 | 32 | 35 |
| 13 | 33 | 33 | 32 | 38 |
| 14 | 31 | 42 | 19 | 45 |
| 15 | 38* | 30* | 34 | 43* |
| 16 | 36 | 34 | 37 | 41 |
| 17 | 40† | 18† | 35 | 14 |
| 18 | 32 | 33 | 32 | 35 |
| 21 | 37 | 37 | 36 | 39 |
| 22 | 38† | 31*† | 34 | 44* |
IVF, in vitro fertilization; MESA, microsurgical sperm aspiration; TESE, testicular sperm extraction.
*p < .025.
†p < .05.
The same analysis was performed in the group of patients with repeated IVF failures. As represented in Table 5, aneuploidy of chromosomes 15, 17, and 22 were significantly lower compared with women in advanced reproductive age; the other frequencies were similar, including that of chromosome 21, although, when analyzed in patients of 36 years and older, this chromosome showed a clear correlation with age (see Fig. 7).
In patients with an altered karyotype due to gonosomal mosaicism, the distribution of the frequencies for aneuploidy did not seem to vary among the different chromosomes, suggesting that, if these patients have a tendency to generate mosaic cell lines, all the chromosomes seem to be equally involved (see Table 7).
Finally, MESA-TESE patients had a pattern similar to that observed in the maternal age group as confirmed by the higher frequencies of chromosomes 15 and 22 (43% and 44%, respectively) in comparison to patients with repeated failures (30% and 31%; p < .025; see Table 7). These results suggest that aneuploidy could be especially frequent in gametes derived from azoospermic patients, as anticipated by the data of the babies born after ICSI, which report an increase in the percentage of de novo chromosomal abnormalities.34
Additional data are needed to confirm these preliminary results, but the main conclusion is that other chromosomes, besides those routinely tested by PGD, could play an important role in determining embryo viability. The correct choice, as far as limitations remain in the number of chromosomes to be analyzed, could strictly depend on the type of patients, their history, and indication for PGD.
PREIMPLANTATION GENETIC DIAGNOSIS FOR TRANSLOCATIONS
The mechanism of chromosome partition followed by reunion in a different configuration is defined as chromosomal translocation. Phenotypical effects are devastating in the case of unbalanced rearrangements due to the altered amount of chromosomal material. By contrast, no loss or gain of genetic material occurs in balanced rearrangements, which generally do not cause clinical consequences except for the production of a high proportion of unbalanced gametes at meiosis. This would originate monosomy or trisomy (even partial) in the resulting embryo and a consequent increased risk of spontaneous abortions and abnormal offspring.
Therefore, carriers of balanced translocations have been proposed to undergo PGD with the aim of improving the clinical outcome by selecting for transfer those embryos with a normal or balanced chromosomal complement. Different technical approaches have been proposed,41,42,43,44 but at present, the most common method to detect reciprocal translocations is based on the simultaneous use of an appropriate combination of telomeric and centromeric probes.45,46 More simple is the case of robertsonian translocations in which enumerator α-satellite or locus-specific probes enable the detection of aneuploid embryos.47
The performance after PGD in carriers of robertsonian and reciprocal translocations is represented in Table 8.
TABLE 8. Preimplantation Genetic Diagnosis in Carriers of Robertsonian and Reciprocal Translocations
| Robertsonian | Reciprocal | |
Cycles (n) | 27 | 16 |
Age (y) (M ± SD) | 35.7 ± 3.7 | 35.0 ± 5.6 |
Embryos FISH diagnosed (n) | 136 | 82 |
FISH normal (n [%]) | 36 (26)* | 10 (11)* |
FISH abnormal (n [%]) | 100 (74)* | 72 (89)* |
Transferred cycles (n [%]) | 16 (59) | 5 (31) |
Transferred embryos (n) (M ± SD) | 1.8 ± 0.8 | 1.2 ± 0.4 |
Clinical pregnancies (n [%]) | 9 (56) | 1 (20) |
Abortions (n) | 2 | 1 |
Implantation rate (%) | 42.9 | 16.7 |
FISH, fluorescence in situ hybridization; M, mean; SD, standard deviation.
*p < .025.
The treatment of 27 carriers of robertsonian translocations demonstrated 26% of embryos with a normal or balanced chromosomal complement; transfer was performed in 16 cycles, generating a clinical pregnancy rate of 56% per transferred cycle and 33% per oocyte retrieval; the overall implantation rate was 42.9% and the take-home-baby rate 26%.
The proportion of FISH abnormal embryos generated by carriers of reciprocal translocations was significantly higher (89%) compared to robertsonian translocations; only 5 cycles in 16 oocyte retrievals had embryos available for transfer with 1 clinical pregnancy. In view of these findings, the number of embryos suitable for chromosomal analysis is obviously crucial for these patients.
The present results confirm the high reproductive risk associated with an altered karyotype. The selection of FISH-normal or balanced embryos has proved to provide good chances of pregnancy, with a significant advantage in terms of ongoing implantation rate compared with a control group in which conventional conception cycles were performed.17 These results are especially relevant when considering that even in the case of embryos at the seven- or eight-cell stage on day 3, chromosomal imbalance was diagnosed in 65%, suggesting that in these categories of patients, normal morphology and development, including blastocyst growth, are not sufficient criteria for chromosomal normality.48 These considerations make PGD for translocations a valuable approach to enhance the chances of a healthy pregnancy in patients who otherwise are destined to experience repeated reproductive failures.
CONCLUSIONS
The data generated worldwide have made PGD an established technique that is offered to patients with a poor prognosis of full-term pregnancy in the hope of improving their reproductive chances.
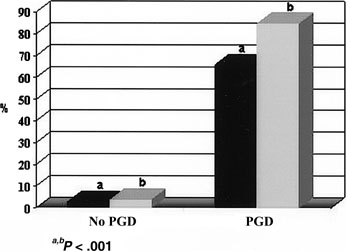
Several studies have been done and others are in progress aimed at demonstrating whether a real advantage derives from the application of PGD compared with conventional treatment. An answer will come soon, but some valuable information is already available, for example, from the analysis of the history presented by 111 on-term pregnancies obtained after PGD. These couples had previously performed 187 cycles obtaining 14 pregnancies, of which 3 went to term and 11 miscarried. They also had had 58 spontaneous pregnancies with 6 delivered and 52 ended in abortion. A calculation was made of the pregnancy and implantation rates derived after PGD and compared with those of conventional treatment cycles; the results are strongly in favor of PGD (Fig. 8). This information cannot be disregarded when pros and cons of PGD are presented to patients, keeping in mind that the final goal is achieving a healthy pregnancy.
REFERENCES
Hassold T, Chen N, Funkhouser J, et al: A cytogenetic study of 1000 spontaneous abortuses. Ann Hum Genet 44:151, 1980 |
|
Warburton D, Stein Z, Kline J, et al: Chromosome abnormalities in spontaneous abortions: Data from the New York City study. In Porter LH, Hook EB (eds): Human Embryonic and Fetal Death. pp 261, 287 New York, Academic Press, 1980 |
|
Warburton D, Kline J, Stein Z, et al: Cytogenetic abnormalities in spontaneous abortions of recognized conceptions. In Porter LH, Willey A (eds): Perinatal Genetics: Diagnosis and Treatment. pp 133, 148 New York, Academic Press, 1986 |
|
Hook EB: Chromosome abnormalities: prevalence, risk and recurrence. In Brock DJH, Rodeck CH, Ferguson MA (eds): Prenatal Diagnosis and Screening. pp 351, 392 Edinburgh, Churchill Livingstone, 1992 |
|
Edwards RG, Gardner RL: Sexing of live rabbit blastocysts. Nature 214:576, 1967 |
|
Handysyde AH, Kontogianni EH, Hardy K, et al: Pregnancies from biopsied human preimplantation embryos sexed by Y-specific DNA amplification. Nature 344:768, 1990 |
|
Verlinsky Y, Ginsberg N, Lifchez A, et al: Analysis of the first polar body: Preconception genetic diagnosis. Hum Reprod 5:826, 1990 |
|
Handysyde AH, John G, Lesko MS, et al: Birth of a normal girl after in vitro fertilization and preimplantation genetic diagnostic testing for cystic fibrosis. N Engl J Med 327:905, 1992 |
|
Munné S, Lee A, Rosenwaks Z, et al: Diagnosis of major chromosome aneuploidies in human preimplantation embryos. Hum Reprod 8:2185, 1993 |
|
Munné S, Alikani M, Tomkin G, et al: Embryo morphology, developmental rates, and maternal age are correlated with chromosomal abnormalities. Fertil Steril 64:382, 1995 |
|
Dailey T, Dale B, Cohen J, et al: Association between non-disjunction and maternal age in meiosis-II human oocytes detected by FISH analysis. Am J Hum Genet 59:176, 1996 |
|
Gianaroli L, Magli MC, Munné S, et al: Will preimplantation genetic diagnosis assist patients with a poor prognosis to achieve pregnancy? Hum Reprod 12:1762, 1997 |
|
Magnuson T, Debrot S, Dimpfl J, et al: The early lethality of autosomal monosomy in the mouse. J Exp Zool 236:353, 1985 |
|
Sandalinas M, Sadowy S, Alikani M: Developmental ability of chromosomally abnormal human embryos to develop to the blastocyst stage. Hum Reprod 16:1954, 2001 |
|
Janny L, Menezo YJR: Maternal age effect on early human embryonic development and blastocyst formation. Molec Reprod Dev 45:31, 1996 |
|
Gianaroli L, Magli MC, Ferraretti AP, et al: Preimplantation genetic diagnosis increases the implantation rate in human in vitro fertilization by avoiding the transfer of chromosomally abnormal embryos. Fertil Steril 68:1128, 1997 |
|
Gianaroli L, Magli MC, Ferraretti AP, et al: Preimplantation diagnosis for aneuploidies in patients undergoing in vitro fertilization with a poor prognosis: Identification of the categories for which it should be proposed. Fertil Steril 72:837, 1999 |
|
Munné S, Magli MC, Cohen J, et al: Positive outcome after preimplantation diagnosis of human embryos. Hum Reprod 14:2191, 1999 |
|
Munné S, Magli MC, Bahçe M, et al: Preimplantation diagnosis of the aneuploidies most commonly found in spontaneous abortions and live births: XY, 13, 15, 16, 18, 21, 22. Prenat Diagn 18:1459, 1998 |
|
Bahçe M, Cohen J, Munné S, et al: PGD of aneuploidy: Were we looking at the wrong chromosomes? J Assist Reprod Genet 16:176, 1999 |
|
Bahçe M, Escudero T, Sandalinas M, et al: Improvements of preimplantation diagnosis of aneuploidy by using microwave hybridization, cell recycling and monocolor labeling of probes. Mol Hum Reprod 9:849, 2000 |
|
Gianaroli L, Magli MC, Munné S, et al: Advantages of day 4 embryo transfer in patients undergoing preimplantation genetic diagnosis of aneuploidy. J Assist Reprod Genet 16:170, 1999 |
|
Gianaroli L, Magli MC, Ferraretti AP, et al: The in vivo and in vitro efficiency and efficacy of PGD for aneuploidy. Mol Cell Endocrinol 183:13, 2001 |
|
Munné S, Marquez C, Magli MC, et al: Scoring criteria for preimplantation genetic diagnosis of numerical abnormalities for chromosomes X, Y, 13, 16, 18 and 21. Mol Hum Reprod 4:863, 1998 |
|
Munné S: Preimplantation genetic diagnosis of numerical and structural chromosome abnormalities. RBM Online 4:183, 2002 |
|
Magli MC, Jones G, Gras L, et al: Chromosome mosaicism in day 3 aneuploid embryos that develop to morphologically normal blastocysts in vitro. Hum Reprod 15:1781, 2000 |
|
Munné S, Dailey T, Finkelstein M, et al: Reduction in signal overlap results in increased FISH efficiency: Implications for preimplantation genetic diagnosis. J Assist Reprod Genet 13:149, 1996 |
|
Magli MC, Sandalinas M, Escudero T, et al: Double locus analysis of chromosome 21 for preimplantation genetic diagnosis of aneuploidy. Prenat Diagn 21:1080, 2001 |
|
Verlinsky Y, Kuliev A: Preimplantation diagnosis of common aneuploidies in fertile couples of advanced maternal age. Hum Reprod 11:2076, 1996 |
|
Gianaroli L, Magli MC, Ferraretti AP, et al: The role of preimplantation diagnosis for aneuploidies. RBM Online 3:31, 2002 |
|
Schatten G: The centrosome and its mode of inheritance: The reduction of the centrosome during gametogenesis and its restoration during fertilization. Dev Biol 165:299, 1994 |
|
Edwards RG, Beard HK: Oocyte polarity and cell determination in early mammalian embryos. Mol Hum Reprod 3:863, 1997 |
|
Liebaers I, Bonduelle M, Van Assche E, et al: Sex chromosome abnormalities after intracytoplasmic sperm injection. Lancet 346:1095, 1995 |
|
Bonduelle M, Aytoz A, Van Assche E, et al: Incidence of chromosomal aberrations in children born after assisted reproduction through intracytoplasmic sperm injection. Hum Reprod 13:781, 1998 |
|
Gianaroli L, Magli MC, Ferraretti AP, et al: Preimplantation diagnosis after assisted reproduction techniques for genetically determined male infertility. J Endocrinol Invest 23:711, 2000 |
|
Simon C, Rubio C, Vidal F, et al: Increased chromosomal abnormalities in human preimplantation embryos after in vitro fertilization in patients with recurrent miscarriage. Reprod Fertil Dev 10:87, 1998 |
|
Gianaroli L, Magli MC, Ferraretti AP, et al: Gonadal activity and chromosomal constitution of in vitro generated embryos. Mol Cell Endocrinol 161:111, 2000 |
|
Kuleshova L, Gianaroli L, Magli MC, et al: Birth following vitrification of a small number of human oocytes. Hum Reprod 14:3077, 1999 |
|
Anderiesz C, Ferraretti AP, Magli MC, et al: Effect of recombinant human gonadotropins on human, bovine and murine oocyte meiosis, fertilization and embryonic development in vitro. Hum Reprod 15:1140, 2000 |
|
Magli MC, Gianaroli L, Ferraretti AP: Chromosomal abnormalities in embryos. Mol Cell Endocrinol 183:29, 2001 |
|
Munné S, Scott R, Sable D, et al: First pregnancies after preconception diagnosis of translocations of maternal origin. Fertil Steril 69:675, 1998 |
|
Munné S, Fung J, Cassel MJ, et al: Preimplantation genetic analysis of translocations: Case-specific probes for interphase cell analysis. Hum Genet 102:663, 1998 |
|
Pierce KE, Fitzgerald LM, Seibel MM, et al: Preimplantation genetic diagnosis of chromosomal balance in embryos from a patient with a balanced reciprocal translocation. Mol Hum Reprod 4:167, 1998 |
|
Verlinsky Y, Evsikov S: Karyotyping of human oocytes by chromosomal analysis of the second polar body. Mol Hum Reprod 5:89, 1999 |
|
Scriven PN, Handyside AH, Mackie OC: Chromosome translocations: Segregation modes and strategies for preimplantation genetic diagnosis. Prenat Diagn 18:1437, 1998 |
|
Munné S, Sandalinas M, Escudero T, et al: Outcome of preimplantation genetic diagnosis of translocations. Fertil Steril 73:1209, 2000 |
|
Conn CM, Harper JC, Winston RML, et al: Infertility couples with robertsonian translocations: Preimplantation genetic analysis of embryos reveals chaotic cleavage divisions. Hum Genet 102:117, 1998 |
|
Evsikov S, Cieslak J, Verlinsky Y: Effect of chromosomal translocations on the development of preimplantation human embryos in vitro. Fertil Steril 74:672, 2000 |